| Hint | Food | 맛과향 | Diet | Health | 불량지식 | 자연과학 | My Book | 유튜브 | Frims | 원 료 | 제 품 | Update | Site |
|
원료 ≫ 식품원료, 첨가물 봉쇄제 Chelating agent 봉쇄제 Chelating agent - 합성품 : EDTA - 유기산 : 피트산, 옥살산 oxalic acid, 구연산, 인산 - 단백질 : 헤모글로빈, CPP, 철분 Siderophores - 미네랄의 흡수조건 : 킬레이트 - 작물의 킬레이트제에 의한 미네랄 흡수 증진 - 중금속 제거 Chelating 요법 - 글리포세이트 : chelate 능력 때문에 위험? Siderophores (Greek: "iron carrier") are small, high-affinity iron-chelating compounds secreted by microorganisms such as bacteria and fungi   • EDTA - 킬레이션제가 항산화제로 작용  • 피트산 phytic acid 쌀(특히 현미), 밀, 옥수수, 콩류와 같은 식물 종자에 다량으로 존재하며 사람, 돼지, 닭, 마우스등에서 생체 활성에 중요한 칼슘, 인, 망간 및 철과 미네랄의 흡수를 방해한다 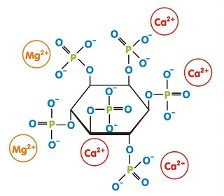 • 옥살산 oxalic acid - 신장결석 : 옥살산, 구연산, 칼슘  옥살산은 킬레이팅력이 강하다. 그래서 칼슘등과 강하게 결합하는데. 신장결석의 주성분이 Calcium oxalate이다. 시금치과 채소와 배추속 식물(양배추,브로콜리,겨자, brussels sprouts)에 많다. 파슬리는 100g당 무려 1.7g의 옥살산이 있어서 시금치(0.97g)보다 1.7배 많다. • 인산 : phosphate Since the work of Schwarzenbach in 1949, phosphonic acids are known as effective chelating agents. NTMP, EDTMP and DTPMP. 이들은 EDTA와 유사구조이다. 인산기가 증가 할수록 킬레이트력이 증가한다. 인산염은 di- and trivalent metal ions을 강하게 결합하여 물의 경도를 낮춘다. 그대서 침전을 방지 할 수 있다. The binding of these ligands also suppresses the catalytic properties of metal ions. They are stable under harsh conditions. For these reasons, an important industrial use of phosphonates is in cooling waters, desalination systems, and in oil fields to inhibit scale formation. Phosphonates are also regularly used in reverse osmosis systems as anti-scalants. Phosphonates in cooling water systems also serve to control corrosion of iron and steel. In pulp and paper manufacturing and in textile industry they serve as "peroxide bleach stabilizers", by chelating metals that could inactivate the peroxide. In detergents they are used as a combination of chelating agent, scale inhibitor, and bleach stabilizer. Phosphonates are also increasingly used in medicine to treat disorders associated with bone formation and calcium metabolism.  • 구연산 citric acid  칼슘은 좋은 킬레이트제로 금속이온을 쉽게 녹게 한다. 보일러 증발기 등의 스케일제거에도 좋으며 물의 경도는 낮추는데도 유용하다. 그래서 구연산은 욕실이나 주방세제에 활성성분으로 사용되기도 한다. 6% 정도의 용액은 묵은 때의 제거에 효과적이다. 녹을 제거하는데도 유용하고, 삼푸에서는 왁스를 녹이는 기능을 한다. 심지어 구연산은 1940년대 원자탄을 개발하기 위한 맨하탄 프로젝트에서 란탄계열의 원자를 분리하는 목적으로도 사용되었다. 그러다 1950년도에는 보다 효과적인 EDTA에 의해 대체되었다 • 유기산 Amino acids, organic acids, glycine betaine, citric acid, and glutamic acid  • 아미노산  • 킬레이트 능력 비교  킬레시션은 흡수도가 달라진다 킬레시션은 치료법도 있다  .jpg)   |
||||
|
|
|||