| Hint | Food | 맛과향 | Diet | Health | 불량지식 | 자연과학 | My Book | 유튜브 | Frims | 원 료 | 제 품 | Update | Site |
|
법칙 ≫ 복합작용 ≫ 상승작용 상승효과 Synergy - 점도 상승효과 : 증점제간의 상호결합 - 감미 상승효과 : 대비효과, sugar, salt - 풍미 상승효과 : sugar, acid, flavor 증점제의 상승작용 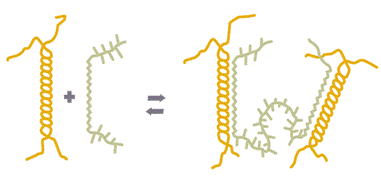  - LBG + Xanthan: 증점제 + 증점제 --> Soft gel --> 강력한 점도 상승작용 - LBG + 카라기난 In many food systems, there are valid reasons for using combinations of hydrocolloids to enhance stability and/or texture. For example, ice cream has a two-part stabilizer system. The primary stabilizer controls the growth rate of ice crystals during the freezing process and slows ice crystal growth during the freeze/thaw cycles experienced during transport and storage. A typical primary stabilizer contains one or more of the following hydrocolloids: LBG, guar gum, cellulose gum, and gelatin. The choice between these gums is based on several factors, including cost-in-use, whether the product is “natural,” whether the product is kosher, and whether freedom from off-flavors is important. The primary stabilizer controls the rate of ice crystal growth, but it also gives the ice cream mix a tendency toward whey separation (“whey-off”). The secondary stabilizer, carrageenan, provides a very slight gel to the mix, which is sufficient to stop whey separation. Table 3-6 is a compatibility chart of the hydrocolloids. This chart should be used only as a general guide because there are exceptions to every rule in food hydrocolloid chemistry. In most cases, there is an additive effect when a gelling gum is used with a thickening gum. Each gum contributes something to the food system, and the end result reflects the two effects. For example, low ester pectin and LBG are used in yogurt fruit preparations. The pectin gives some gel structure (yield point) while the LBG gives some body (viscosity) and controls syneresis. In some cases, the combination of two different thickening gums gives more viscosity than predicted. For example, if a 1% cellulose gum solution with a viscosity level of 4,000 cP is mixed with an equal amount of a 1% solution of guar also having a viscosity level of 4,000 cP, the net result is not 4,000 cP for the mixture. It is, in fact, closer to 6,000 cP. This provides an economic incentive to use synergistic gum systems, because the overall level of gum use is lower. Similarly, xanthan gum gives a synergistic viscosity increase to cellulose gel and cellulose gum. However, one must be sure to specify cellulasefree xanthan gum, because it normally does contain a small amount of cellulase enzyme, which quickly degrades the cellulose gum with a resultant loss in viscosity. In a few cases, a synergistic gelation occurs when two hydrocolloids are combined. The best-known example of this is a mixture of LBG and xanthan gum. If LBG and xanthan are placed together in water at sufficient concentration, heated to 70°C (158°F), and allowed to cool, a rubbery but rigid gel results. As little as one part LBG in 500 parts xanthan (or vice versa) creates a gel if their total concentration in water is 0.5–1%. Similarly, xanthan gum and Tara gum gel when combined. There are cases of a gum changing the gel strength of another gum. For example, agar has less gel strength in the presence of sodium alginate or (to a lesser extent) gum karaya. However, agar has more gel strength in the presence of LBG or (to a lesser extent) cellulose gum. LBG greatly modifies the texture of a κ-carrageenan gel, making it more elastic with a higher break strength, less brittle, and less prone to syneresis. Also, cellulose gel has more viscosity but less thixotropy in the presence of too much CMC, MC, MHPC, or HPC. Some other unusual but potentially useful interactions include the following: 1. Gels made with sodium alginate and high ester pectin are thermally reversible, whereas gels of alginate alone or high ester pectin alone are usually not thermally reversible. The gel temperature of the algin–pectin gel mixture increases as the system’s pH is lowered. 2. Xanthan gum reduces the reaction of low ester pectin to calcium. Thus, more calcium is required to gel low ester pectin in the presence of xanthan gum. 3. Gellan gum has been used in combination with low ester pectin to “extend” the low ester pectin (i.e., to reduce the use level of the pectin without sacrificing gel strength). 4. Pectin and gelatin combinations are additive at pH values above 4.5 or below 3.3. At pH 3.4–4.4, precipitation occurs, unless the system has a high soluble solids content (75% or higher). 5. Low-concentration mixtures of gum arabic and gelatin precipitate and form a coacervate, which is used by the flavor industry for encapsulation. Higher concentrations of the two are used by the confectionery industry to make a pastille with a soft, rubbery texture. In this case, there is no precipitation because of the crowded hydration conditions and the resulting lower water activity. 6. Mixtures of gelatin and carrageenan increase gel strength synergistically in food systems at pH 6.0 or higher. Gelatin and gellan gum show a similar increase in synergistic gel strength at pH values above 5.0. At acid pH values (below 5), gelatin precipitates either of these two anionic hydrocolloids. 7. As a general rule, gelatin is compatible and potentially synergistic with anionic hydrocolloids when it is negatively charged (pH values above the isoelectric pH) and coprecipitates with anionic hydrocolloids when it is positively charged (pH values below the isoelectric pH). In other words, gelatin can be used only in combination with anionic hydrocolloids in neutral or near-neutral pH systems, such as milk or meat products. The exception to this rule is systems with low water activity and low pH, such as certain confectionery formulations where gum arabic–gelatin or pectin–gelatin combinations are used to attain specific textures. |
||||
|
|
|||