| Hint | Food | 맛과향 | Diet | Health | 불량지식 | 자연과학 | My Book | 유튜브 | Frims | 원 료 | 제 품 | Update | Site |
|
원료 ≫ 산미료 산미료 종류 산미료 종류 - 염산, 질산, 황산, 인산, 탄산 - 1 : 초산 , 프로피온산, 젖산 - 2 : 호박산Succinic acid, fumaric acid, 사과산malic acid, 주석산tartaric acid - 3 : 구연산 Citric acid - 락톤 : Ascorbic acid, gluconolactone - 페놀산: Benzoic acid - 지방산 : Sorbic acid, caprylic acid, butyric - 아미노산 - 옥살산 oxalic acid, 피트산 , 포름산 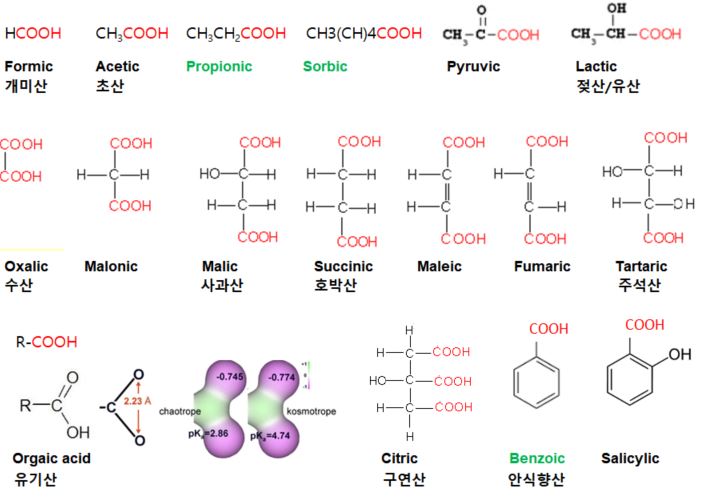 ① 구연산(결정, Citric Acid) 구연이란 시트론 citron의 한자명이며, Citrus 과일에 특히 많이 함유되어 있는 데에서 연유 무색의 투명한 결정, 알맹이 또는 덩어리거나 백색의 결정성 분말 무취, 신맛. 물, 알콜에 용해도 좋음. 모든 유기산중에서 가장 부드럽고 상쾌하여 산미료의 표준. ② 구연산(무수, Citric acid Anhydrous) 구연산의 무수물로서 무색투명한 결정, 알맹이 또는 백색의 분말, 신맛은 결정구연산의 약 1.1배 흡습성이 적으며 잘 굳어지지 않음. Solubility in water 133 g/100 ml (22°C) Acidifier, curing accelerator, dispersing agent, flavoring agent, sequestrant, and synergist antioxidant Widely used in sherbet, ice cream, ices, beverages, salad dressing, fruit preserves, jams and jellies Used as acidulant in canned vegetables Calcium citrate is used for firming potatoes, and tomatoes during processing Important acidulant fo dairy products because it is precursor of diacetyl therefore improves the flavor and aroma of cultured dairy products Major acidulant in carbonated drink and as preservative in syrup and beverages Retards browning reaction Chelating agent ⑦ 구연산나트륨(Sodium Citrate) 무색의 결정 또는 백색의 결정성 분말로 냄새가 없으며 상쾌한 소금 맛 물에는 잘 용해되나 알콜에는 용해되지 않음. 수용액은 약한 알칼리성이며 5% 용액의 pH는 7.6 ~ 8.6 사용기준은 없으며 식품에 완충작용, 유화 안정작용에 널리 사용. 청량음료 등에 구연산을 사용할 때 산미를 완화할 목적으로 구연산과 같이 첨가. ③ D-주석산(D-Tartaric acid) 포도주를 만들 때 침전하는 주석(酒石)에 함유되어 있어 주석산 (또는 포도산) 무색 투명한 결정 또는 백색의 미세한 결정성 분말, 무취. 구연산에 비해 신맛은 1.2 ~ 1.3배 강. 다소 떫은 맛, 상쾌감. 공기나 광선에 대해서 안정. ④ DL-주석산(dl-Tartaric acid) 무색의 결정 또는 백색의 결정성 분말이고 냄새는 없으며 신맛 물에 대한 용해도가 D-주석산보다 낮고 불용성인 칼슘염이 생성되기 쉬우므로 사용상 주의가 필요. 흡습성도 극히 적다. Solubility in water 133 g/100ml (20°C) Used as acidifier, firming agent, flavor enhancer, flavoring agent, humectant, pH control agent, and sequestrant Tartaric acid is used in lime and grape flavored beverages Commonly ingredient in baking powder and leavening systems The limited solubility of tartaric acid in water prevent premature leavening as required in dough mixing stage Choline bitartaric is used as dietary supplement ⑤ 푸말산(Fumaric acid) 백색의 결정성 분말로 냄새는 없고 특이하고 상쾌한 신맛 승화성. 물에 잘 녹지 않는다. Solubility in water 0.63 g/100 mL Polyfunctional chemical Impart a sour taste of food One of the most acidic solid acid Acidifier, curing accelerator, and flavoring agent Used extensively in fruit juice drink, gelatin dessert, pie filling, refrigerated biscuit dough, and wines Used in preparing edible coating for candy Has good antioxidant properties ⑥ DL-사과산(DL-Malic acid) 백색의 결정성 분말 또는 백색의 결정. 냄새는 없거나 약간의 특이한 냄새가 있으며 특이하고 약간 자극적인 신맛. 물에 대한 용해도는 구연산과 거의 비슷. 청량음료수, 젤리, 젖산균음료등에 보통 다른 유기산과 병용. Solubility in water 558 g/l (at 20 °C)[1] Predominant acid in apples, apricots, bananas, cherries, grapes, orange peels, peaches, pears, and plums Used as acidifier, flavor enhancer, flavoring agent, pH control agents, and synergist for antioxidant Less malic acid than citric acid is required to impart the same degree of acidity - 개미산 (formic acid) 1670년 피셔가 개미를 증류하여 처음으로 얻어 개미산이라고 함 - 호박산 (succinic acid) 1550년 R.아그리콜라가 화석(化石)이 된 수지인 호박(琥珀)을 건류하여 얻었다는 기록이 있는 데서 호박산이라고 Normal constituent of plant and animal tissue Flavor enhancer, miscellaneous and general purpose food chemical, neutralizing agent, pH control agent Reaction of succinic acid with protein are often used for modifying plasticity of bread dough The derivatives of succinic acids are used for flavoring agents - 낙산 (butyric acid) 버터 등 동물의 유지방 속에 함유되어 있어서 낙산(酪酸)이라고 한 것이 아닐까 싶습니다. 저 酪자는 "타락 낙(락)"자인데 타락이란 우유를 끓여만든 음료라고 하는군요. - 초산 (acetic acid) 식초의 신맛을 내는 성분이죠. Colorless, waterlike liquid, vinegary odor, and burning taste Used as acidifier, flavor enhancer, flavoring agent, pH control agent, pickling agent, solvent, and antimicrobial Extensively used in preparation of salad dressing, mayonaise, sour and sweet pickles, sauces, cheese, chewing um, dairy products, baked goods, curing meat, canning certain vegetables, infant feeding formula to replace lactic acid More effective in limiting yeast and bacterial than mold growth - 젖산, 유산 (lactic acid) 우유가 발효되면 생기는 산입니다. 요즘엔 우리말로 젖산, 발효균을 젖산균 The most widely distributed organic acid in nature The earliest food additives Used as acidifier, antimicrobial agent, curing agent, flavor enhancer, flavoring agent, pH control agent, and pickling agent Used in manufacture of jams, jellies, confectionery, sherbet, and beverages It is the preferred acidulant fr adjusting acidity amd ensuring the clarity of brines for pickles and olives Used in frozen desserts to provide mild and tart flavor The calcium of lactic acid is primarily used to preserve the firmness of apple slice, inhibit discoloration, gelling agent for dehydrated pectines, improve properties of dried milk powder ASCORBIC ACID Antimicrobial and antioxidant An adjunct to meat curing system Ascorbate or isoascorbate reduce nitrite, forming dehydroascorbic and nitric oxide The later reacts with myoglobin under reducing condition to yield nitrosomyoglobin Ascorbic acid accelerates color development and promotes color uniformity and stability Ascorbis acid and its sodium and calcium salts are used as nutritive additives Used as acidulant to adjust pH to prevent enzymatic browning of fruits and vegetables 품 종 구연산(%) 사과산(%) 오렌지 navel 0.54-0.93 0.05-0.20 Valencia 0.98 0.16 자몽 1.19-2.10 0.04-0.06 레몬 4.00-4.38 0.17-0.26  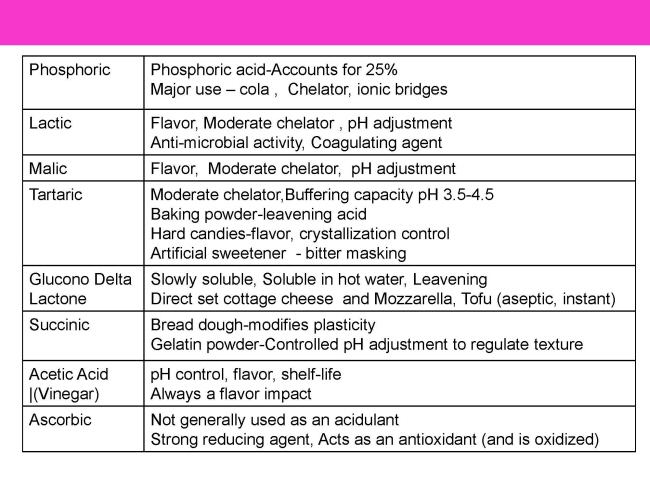 PROPIONIC ACID AND ITS SALTS Liquid with slightly pungent and diagreeable rancid odor Its salts are white, free-flowing powders with a cheese like flavor Propionic acid and its salts are preservatives Used primarily in baked products to suppress bacteria causing rope in the center of the bread and growth of mold on both bread and cakes Also acts as mold inhibitor in cheese foods and spreads Added to bread dough without interfering with leavening and yeast ADIPIC ACID Flavoring agent, leavening agent, neutralizing agent, and pH control agent Limitation on its use: 0.05% in baked goods, 0.005% in nonalcoholic beverages, 5.0% in condiments, 0.45% in dairy products, 0.3% in fats and oils, 0.0004% in frozen dairy dessert, 0.55% in gelatin and pudding, 0.1% in gravies, 0.3% in meat products, 1.3% in snack foods, and 0.02% in other food categories Imparts smooth, tart taste to foods Extensively used in gelatin dessert and liquid and powder beverages As leavening acidulant in baking powder, candies, and refrigerated roll Improves melting characteristics and texture of processed cheese and cheese spread Increase whipping quality of products containing egg white Gel inducing agent in imitation jams and jellies As sequestrant in edible oils 염산 Hydrocloric acid Rarely used as an acidulant, it finds many application in the food industry Permitted as acidulant by the FAO (1974) Used to produce the chloride salts of several important food additives Used in process that require hydrolysis of starting materials such as proteins and starches It is used in production of corn syrup 황산 Sulfuric Acid It is not directly used as acidulant The most important inorganic acid Finds several application in the manufacture and synthesis of various additives used in food Used for hydrolysis The various sulfate derivatives used in food application are also prepared using sulfuric acid 
|
||||
|
|
|||